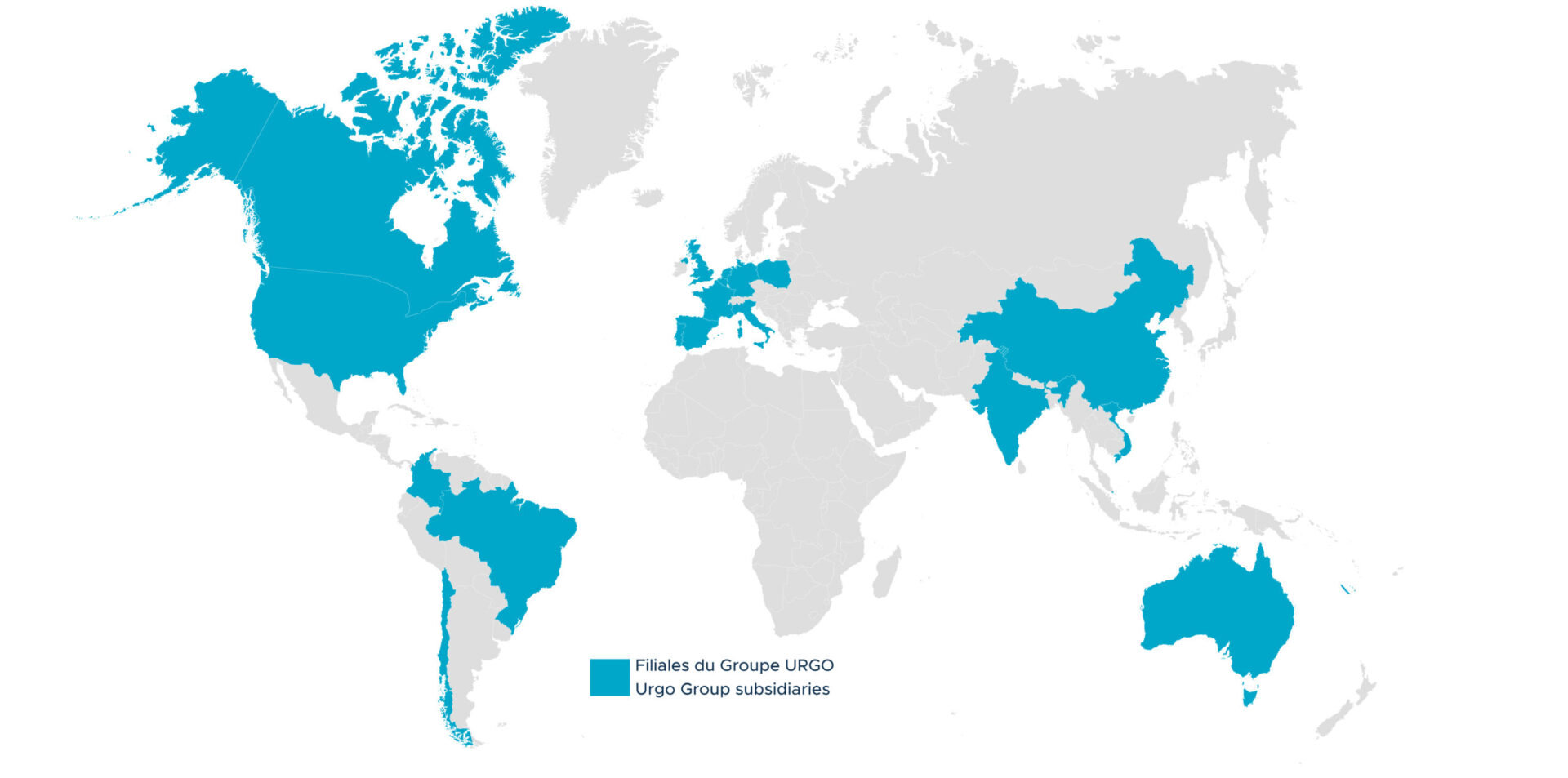
Since 2011, the URGO Group has banked on international business in its development strategy. It now generates half of its turnover outside France.
Our presence in 20 countries through subsidiaries or strategic partnerships allows us to offer our staff a variety of career paths.